The acquisition of Bellwyck forms part of PCI’s plan to expand global clinical trial services into Canada and Continental Europe
PCI Pharma Services has announced the acquisition of Bellwyck Pharma Services. (Credit: PCI Pharma Services)
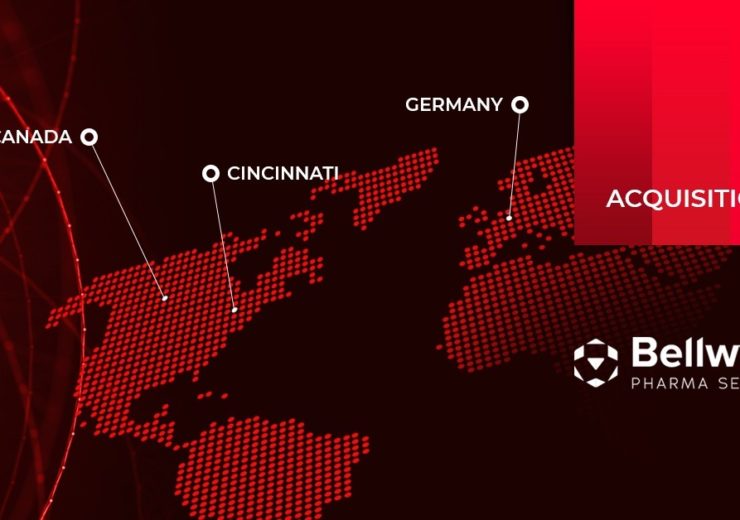
Integrated pharmaceutical development services provider PCI Pharma Services (PCI) has acquired Bellwyck Pharma Services (Bellwyck), a Canada-based provider of packaging and labelling for clinical trial and commercial drug markets.
The acquisition is a part of PCI’s efforts to expand global clinical trial services into Canada and Continental Europe. Financial terms of the deal were undisclosed.
Bellwyck operates four GMP-compliant facilities, which will add to PCI’s network in the US, Germany and Canada.
PCI Pharma Services CEO Salim Haffar said: “We are thrilled to welcome the Bellwyck team into the PCI family.
“Their passion for patients, talented team and innovative approach to client challenges is perfectly aligned to our purpose and global strategy as PCI strives to be the bridge between life-changing therapies and patients around the world.
“We are particularly excited that this acquisition will expand our clinical business worldwide and provide our clients with access to sites in continental Europe and Canada, in addition to our existing facilities.”
PCI said that the clinical and commercial supply chain solutions of Bellwyck reflect its existing core capabilities including clinical trial packaging, labelling and logistics for Phase I-IV studies as well as full-service commercial packaging.
PCI’s total number of GMP-compliant facilities increased to 25
The acquisition brings the PCI’s total number of GMP-compliant facilities to 25 across North America, Europe and Asia-Pacific.
Bellwyck Pharma Services CEO Jeff Sziklai said: “We have a shared focus on innovation, quality, exceptional service and our employees. Joining forces with a global industry leader, such as PCI, is a great step forward for Bellwyck.
“We look forward to helping accelerate the global growth of PCI’s clinical business, leveraging our reputation for innovative, customized clinical trial solutions for our clients.”
In February 2019, PCI Pharma Services has invested to boost its bottling line capacity at its commercial packaging site in Rockford, Illinois.
The investment at the Rockford location is a part of PCI’s series of capacity expansion initiatives across its worldwide network.
