ActiveGuard Connect smart packaging solutions will help boost medication adherence by 23%
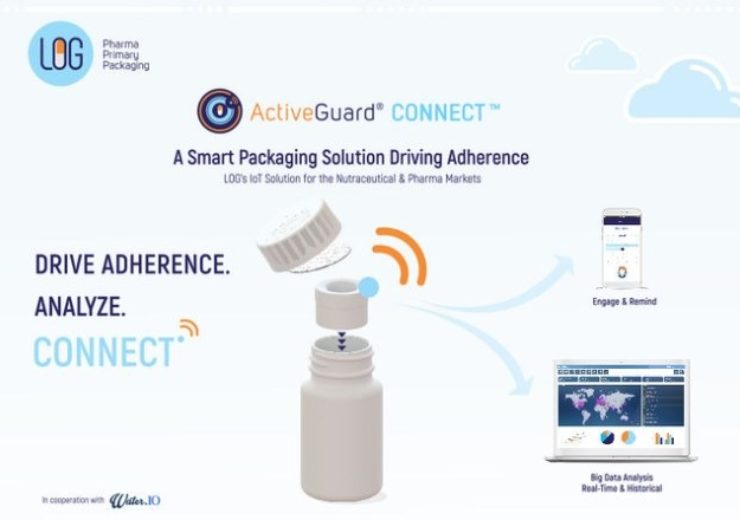
Image: With ActiveGuard Connect, LOG is providing the pharma and nutraceutical industries with a smart packaging solution driving adherence.
LOG Pharma Packaging has unveiled its patent-pending line of ActiveGuard Connect smart packaging solutions.
LOG Pharma, in collaboration with Water.IO, has developed ActiveGuard Connect to address the lack of patient adherence to prescription medicine or supplementation of nutraceuticals.
ActiveGuard Connect will help indicate taken doses and track temperature within the bottle
ActiveGuard Connect is provided with a smart IoT component with integrated desiccant chamber, which links and synchronises with a patient’s smartphone.
The smart packaging product facilitates indication of taken doses and tracks temperature within the bottle, and reminds or alerts patients when required to drive adherence.
The company’s smart packaging solution facilitates Big Data analysis based on historical and real-time data to track compliance, and helps pharma and nutraceutical manufacturers to improve medication and supplement efficiency.
ActiveGuard Connect also offers manufacturers a unique connection to engage with the end-customer and develop a long-term relationship while increasing sales.
The company is exhibiting the ActiveGuard Connect smart packaging solutions at this year’s CPhI Worldwide event in Frankfurt, Germany, being held from 5 to 7 November.
LOG Pharma Packaging CEO Ephraim Ulmer said: “With only 30-50% medication adherence rate within the general population, ActiveGuard® CONNECT™ harnesses IoT technology and innovation to deliver a patient-centric solution. Using smart packaging and active customer engagement has been proven to increase adherence by 23%.
“We are proud to add ActiveGuard® CONNECT™ to LOG’s line of high-quality innovative and smart packaging products for the nutraceutical and pharmaceutical markets.”
Founded in 1971, LOG Pharma Packaging provides advanced primary packaging solutions to global pharmaceutical companies.
The company provides a range of packaging solutions to deliver both tablets and liquid formulations in better condition.
In addition, the firm produces a range of advanced active and passive barrier solutions, which minimise drug formulation development time, reduce time-to-market (TTM), extend shelf life and help in passing drug stability tests.
