Both firms have developed a Proof of Concept (PoC) to provide primary packaging with a unique ID to access its own trustworthy digital twin
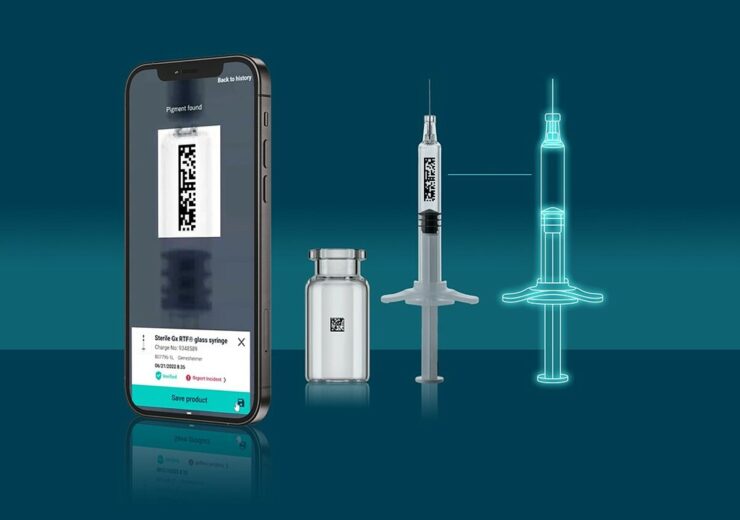
Digital twins ensure digital authenticity and trust through patented crypto anchors and digital certificates. (Credit: Gerresheimer AG)
Drug delivery systems maker Gerresheimer and science and technology company Merck have jointly developed a digital twin solution to further ensure traceability and trust in the pharmaceutical supply chain.
Both firms have developed a Proof of Concept (PoC) to provide primary packaging with a unique ID to access its own trustworthy digital twin. The PoC aims to transform the pharmaceutical supply chain by creating digital twins for primary packaging.
The automated process will result in full traceability and digital trust, resulting in cost savings, improved quality and even new business models through a single interconnected network.
Gerresheimer digitalisation and new business models vice president Daniel Diezi said: “We are convinced that our primary packaging will become the key to enable supply networks across organisations and lead to faster process operations and data-informed decision making.”
Gerresheimer said that by using the jointly created solution, the physical primary packaging for Gerresheimer is given a reliable “key” that grants access to its digital twin.
The primary packaging which includes syringe and vial are connected to their individual ID to travel across the whole supply chain, from production to the point of care, linking and gathering data. They turn into the key to unblocking data from multiple digital ecosystems, enabling more effective communication between diverse supply network firms.
According to Gerresheimer, the new approach enables physical objects to be securely anchored in the digital world using a blockchain-based platform and multi-patent authentication technologies created by Merck.
When combined with Industry 4.0 standards, the process becomes more trustworthy and dependable.
The joint PoC between Gerresheimer and Merck includes a smartphone app, real syringes with a secure unique ID, and access to Merck’s digital platform.
Authorised stakeholders, such as brand owners (holders of marketing authorisations) and quality assurance officers of the company, can access the smartphone app.
The authorised user can access services like quality data on demand, root-cause analysis data, and customer complaint handling after authenticating the syringe and reading the unique ID.
